Welcome to SoiLab
Soilab is DIY focused YouTube channel with the goal of creating engaging, easy to understand and entertaining content around soils and plant health. We look to bring the “why” behind your everyday lawncare and garden management decisions. To support these decisions, we will conduct trials and replicated experiments that scientifically back the decision processes. Having access to a full production laboratory, we have a unique ability to test and evaluate complex scientific concepts in a controlled environment, allowing the viewer to grow and better understand soil and plant science. If there is something you wish to see, please reach out to us for to incorporate into future videos! See you in the Lab!
What is Gypsum and How Does It Help My Lawn & Garden
First, what is gypsum? Chemically, gypsum is calcium sulfate (CaSO4); functionally gypsum can be seen as a soil amendment, soil conditioner, or fertilizer. A soil amendment is anything worked into the soil to make it a more hospitable environment for plant growth and development (gypsum, synthetic conditioner, sawdust, or other organic material), while a soil conditioner is added specifically for the purpose of enhancing physical soil properties such as soil structure.
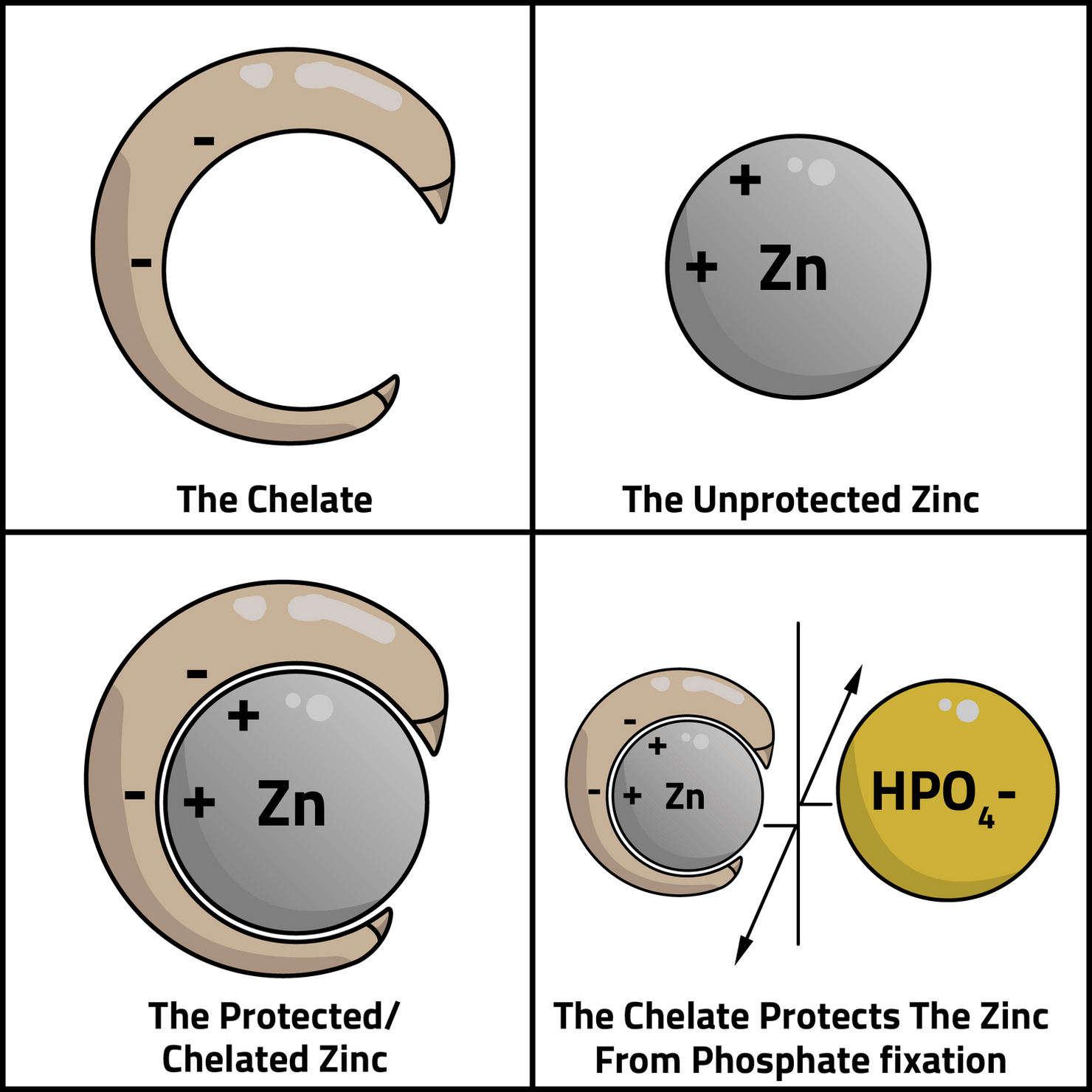
Chelated Zinc Fertilizers
So, you got your soil test results back and it recommended and application of a micronutrient such as copper, iron, manganese or zinc. Now it’s time to pick a product. You’ll likely notice that there are many options and many different price points for the same amount of fertilizer. Why is this? There are several factors at play here, but after inspecting the labels of these fertilizers a bit closer you’ll likely recognize that some may contain, for example, Zinc Sulfate, while others may contain Zinc EDTA, Zinc DTPA, Zinc EDTA & IDS, or Zinc EDDHA and so on. The last four listed indicate that the zinc is coupled with a chelating agent, or in other words, that the zinc is chelated. The word ‘chelate’ is derived from the Greek word for claw or lobster claw. What is the difference between a nutrient that is chelated and one that isn’t? In the case of Zinc and many other micronutrients, once applied to soil in their elemental form they are susceptible to being fixed, or made unavailable, by different chemical and biological processes. A great example is that when Zinc is applied to soils with pH greater that 6.5, especially those higher than 7.0, it is susceptible to being fixed by phosphate, making it unavailable. When Zinc Sulfate is added to the soil, the following reactions are likely to occur, making some of the applied zinc unavailable for plant uptake.
SoiLab pH Trial One Month Update
Here at MySoil we often get questions from DIYers about soil pH adjustments, both up and down. What should I apply? How much? How long does it take to work? How does it work? To help answer these questions we invited Matt from SoiLab to design and implement a pH adjustment trial that will perform 240 MySoil tests to ensure replicated and reliable data. Remember that the soils used in this trial are just a small sample, but directional trends should be similar to your own application. Depending on the unique chemistry of each individual soil, the time it takes to adjust a soils pH will vary. Keep amending and monitoring until you find the “sweet spot”.